














The Sanitary Stainless Steel Solution Preparation Unit is a specialized pharmaceutical apparatus designed for liquid mixing and dispensing operations. This equipment enables the preparation of pharmaceutical solutions by combining active pharmaceutical ingredients (APIs) with solvents such as water for injection. Through integrated processes including controlled stirring, heating, sterilization, and filtration, the system produces medicinal solutions at precise target concentrations for both sterile and non-sterile environments.


Pre-tested modular equipment reduces installation costs, accelerates delivery, and ensures operational flexibility.

Features 1-click CIP/SIP with auto-detection. Includes computerized recipe management, e-signatures, and full audit trails for compliance.
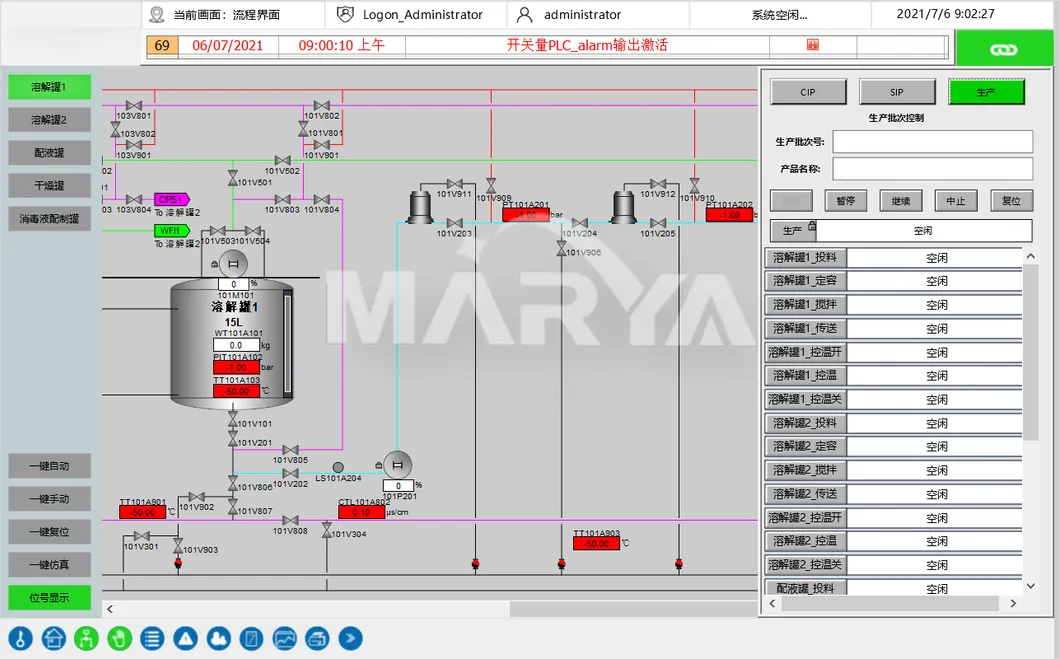
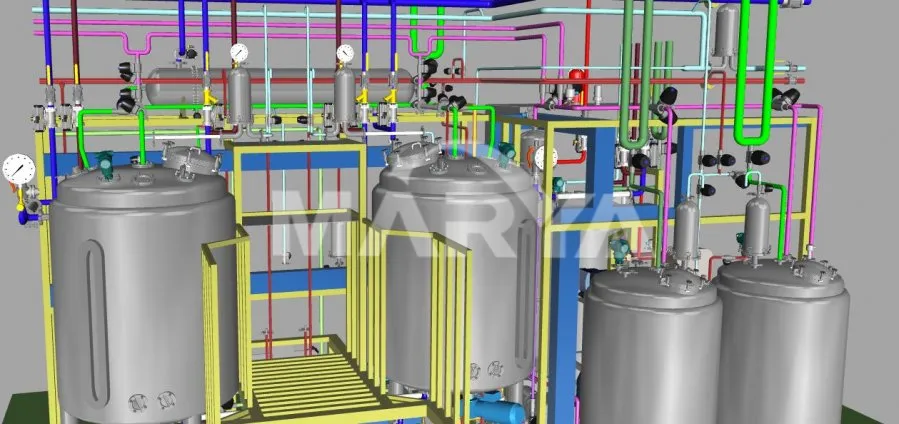
Critical parameter control (temp, DO, pH) with auto-alerts. Pre-production 3D modeling optimizes piping and ergonomic layout.

| Feature Name | Parameters |
|---|---|
| Working Volume | 2 ~ 15,000L |
| Material | 304, 316L, 904L, TA2 |
| Stirring Type | Upper mechanical / Lower magnetic stirring |
| Speed Accuracy | Range 0~450rpm (±3.0%) |
| Temperature Accuracy | Range -10~150ºC (±0.2ºC) |
| Pressure Accuracy | Range -0.01Mpa~0.06Mpa (±0.01MPa) |
| Weighing Accuracy | 3‰ |
| Protection Level | IP65 |










The system is highly scalable, offering working volumes ranging from small-scale 2L units up to large industrial 15,000L tanks.
We primarily use 316L stainless steel for contact parts to ensure pharmaceutical grade hygiene, but options like 304, 904L, and TA2 are also available based on requirements.
Yes, the unit features fully automated 1-click CIP (Clean-in-Place) and SIP (Sterilize-in-Place) operations with automatic detection of cycle completion.
The equipment supports a wide temperature range from -10ºC to 150ºC with a high precision control accuracy of ±0.2ºC.
Absolutely. The units are designed to meet CE, FDA, GMP, and ISO standards, ensuring suitability for global sterile and non-sterile pharmaceutical manufacturing.
The system offers both upper mechanical stirring and lower magnetic stirring options, both equipped with variable frequency speed regulation for precise mixing.